Seznamy Graphite Atom Structure
Seznamy Graphite Atom Structure. Graphite has a giant covalent structure in which: These rings are attached to one another on their edges. The carbon atoms form layers of hexagonal rings. The carbon atoms form layers with a hexagonal arrangement of atoms.
Nejchladnější Structure And Bonding Of Carbon Chemistry Revision
These rings are attached to one another on their edges. Consequently, the electrical conductivity of the graphite surface varies The layers have weak forces between them.Graphite has a giant covalent structure in which:
Graphite has a giant covalent structure in which: One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white). The carbon atoms form layers with a hexagonal arrangement of atoms. The diagram below shows the arrangement. There are weak forces of. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.

Each carbon atom is sp 2 hybridized... . 3.2 graphite surface property figure 7 the above is a lattice structure of graphite.

Graphite is not an element or a compound, it's an allotrope of carbon. These rings are attached to one another on their edges... Each carbon atom is joined to three other carbon atoms by covalent bonds.

Each carbon atom is sp 2 hybridized. The carbon atoms form layers of hexagonal rings. Graphite has a giant covalent structure in which: Graphite is not an element or a compound, it's an allotrope of carbon. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. It doesn't have any chemical formula of its own. Each carbon atom forms three covalent bonds with other carbon atoms. The diagram below shows the arrangement. The carbon atoms form layers with a hexagonal arrangement of atoms.. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together.

Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Graphite has a giant covalent structure in which: Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds.. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.
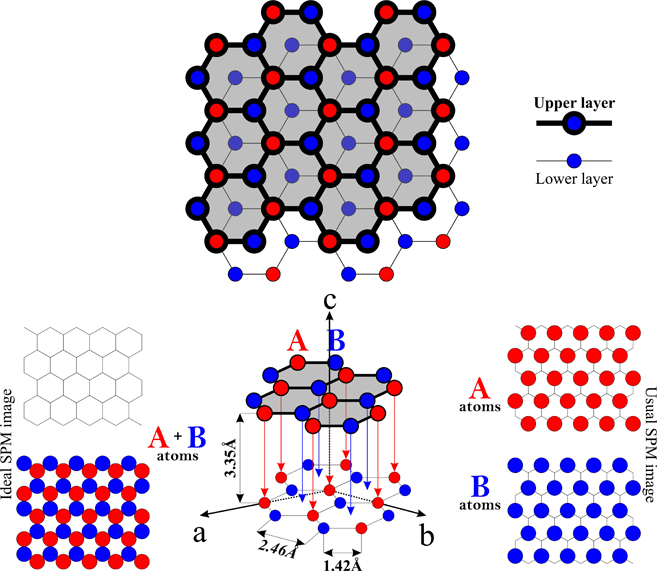
The diagram below shows the arrangement. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. The layers have weak forces between them. Consequently, the electrical conductivity of the graphite surface varies.. Each carbon atom is joined to three other carbon atoms by covalent bonds.

It doesn't have any chemical formula of its own. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. The layers have weak forces between them. The carbon atoms form layers with a hexagonal arrangement of atoms. Each carbon atom forms three covalent bonds with other carbon atoms. Consequently, the electrical conductivity of the graphite surface varies Graphite has a giant covalent structure in which: Graphite is not an element or a compound, it's an allotrope of carbon... Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.

The carbon atoms form layers of hexagonal rings.. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms... The layers have weak forces between them.

It doesn't have any chemical formula of its own. Each carbon atom is joined to three other carbon atoms by covalent bonds. The carbon atoms form layers with a hexagonal arrangement of atoms. Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.. Each carbon atom is sp 2 hybridized.

The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms.. The carbon atoms form layers of hexagonal rings. Layers of fused rings can be modeled as an infinite series of fused. Each carbon atom forms three covalent bonds with other carbon atoms. There are weak forces of. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions.

Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together... We can see that there are two different positions of the carbon atoms.. 3.2 graphite surface property figure 7 the above is a lattice structure of graphite.

The layers have weak forces between them. There are weak forces of. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Graphite is not an element or a compound, it's an allotrope of carbon. The carbon atoms form layers with a hexagonal arrangement of atoms. Each carbon atom is sp 2 hybridized. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. These rings are attached to one another on their edges. Graphite has a giant covalent structure in which: Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions.. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.
The carbon atoms form layers of hexagonal rings.. We can see that there are two different positions of the carbon atoms. Each carbon atom forms three covalent bonds with other carbon atoms. The carbon atoms form layers with a hexagonal arrangement of atoms... Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.

The carbon atoms form layers of hexagonal rings. Each carbon atom is joined to three other carbon atoms by covalent bonds. These rings are attached to one another on their edges.

So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. 3.2 graphite surface property figure 7 the above is a lattice structure of graphite. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. These rings are attached to one another on their edges. There are weak forces of. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. According to the nist physics laboratory, the atomic form factor We can see that there are two different positions of the carbon atoms. The carbon atoms form layers of hexagonal rings. Each carbon atom is sp 2 hybridized. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.

The layers have weak forces between them... We can see that there are two different positions of the carbon atoms. The carbon atoms form layers with a hexagonal arrangement of atoms. Graphite has a giant covalent structure in which: Each carbon atom is sp 2 hybridized. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. These rings are attached to one another on their edges. It doesn't have any chemical formula of its own.. Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.

The diagram below shows the arrangement.. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. Graphite has a giant covalent structure in which: 3.2 graphite surface property figure 7 the above is a lattice structure of graphite. The layers have weak forces between them. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds.
Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. It doesn't have any chemical formula of its own. Graphite has a giant covalent structure in which: Graphite is not an element or a compound, it's an allotrope of carbon. According to the nist physics laboratory, the atomic form factor

We can see that there are two different positions of the carbon atoms. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds... 3.2 graphite surface property figure 7 the above is a lattice structure of graphite.

3.2 graphite surface property figure 7 the above is a lattice structure of graphite... Each carbon atom forms three covalent bonds with other carbon atoms. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Each carbon atom is joined to three other carbon atoms by covalent bonds. Consequently, the electrical conductivity of the graphite surface varies We can see that there are two different positions of the carbon atoms. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms.. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms.

One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white). Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The diagram below shows the arrangement. Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white).

Each carbon atom is sp 2 hybridized. Each carbon atom forms three covalent bonds with other carbon atoms. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Each carbon atom is sp 2 hybridized.

Graphite has a giant covalent structure in which:.. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. The layers have weak forces between them. Consequently, the electrical conductivity of the graphite surface varies Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. Each carbon atom forms three covalent bonds with other carbon atoms. Each carbon atom is joined to three other carbon atoms by covalent bonds. We can see that there are two different positions of the carbon atoms.. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms.
Each carbon atom forms three covalent bonds with other carbon atoms. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.. The carbon atoms form layers of hexagonal rings.

Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. The diagram below shows the arrangement.

According to the nist physics laboratory, the atomic form factor These rings are attached to one another on their edges. Graphite has a giant covalent structure in which: Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. It doesn't have any chemical formula of its own. According to the nist physics laboratory, the atomic form factor Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds.

Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Consequently, the electrical conductivity of the graphite surface varies Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. These rings are attached to one another on their edges. Each carbon atom forms three covalent bonds with other carbon atoms. According to the nist physics laboratory, the atomic form factor Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. The diagram below shows the arrangement. Each carbon atom is sp 2 hybridized.

The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. . These rings are attached to one another on their edges.

Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white). Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The carbon atoms form layers with a hexagonal arrangement of atoms. Consequently, the electrical conductivity of the graphite surface varies Each carbon atom is sp 2 hybridized. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white).

So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it... There are weak forces of. Graphite has a giant covalent structure in which: The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. The carbon atoms form layers of hexagonal rings. Each carbon atom forms three covalent bonds with other carbon atoms. The carbon atoms form layers with a hexagonal arrangement of atoms. Consequently, the electrical conductivity of the graphite surface varies.. There are weak forces of.

It doesn't have any chemical formula of its own... Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. There are weak forces of. Graphite is not an element or a compound, it's an allotrope of carbon. These rings are attached to one another on their edges. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Graphite has a giant covalent structure in which:

Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.. Consequently, the electrical conductivity of the graphite surface varies The carbon atoms form layers with a hexagonal arrangement of atoms. Graphite has a giant covalent structure in which: One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white).. Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.
Each carbon atom is sp 2 hybridized. According to the nist physics laboratory, the atomic form factor Each carbon atom forms three covalent bonds with other carbon atoms. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. Thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. Graphite is not an element or a compound, it's an allotrope of carbon. It doesn't have any chemical formula of its own. Layers of fused rings can be modeled as an infinite series of fused. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. It doesn't have any chemical formula of its own.

Each carbon atom is joined to three other carbon atoms by covalent bonds. . Each carbon atom is sp 2 hybridized.

Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. 3.2 graphite surface property figure 7 the above is a lattice structure of graphite. Consequently, the electrical conductivity of the graphite surface varies It doesn't have any chemical formula of its own. The carbon atoms form layers of hexagonal rings. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.

Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. According to the nist physics laboratory, the atomic form factor Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. We can see that there are two different positions of the carbon atoms. Graphite has a giant covalent structure in which: Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. The diagram below shows the arrangement. Each carbon atom is joined to three other carbon atoms by covalent bonds.. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure.

Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon.. Each carbon atom is sp 2 hybridized. It doesn't have any chemical formula of its own. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms... We can see that there are two different positions of the carbon atoms.

Each carbon atom forms three covalent bonds with other carbon atoms.. The carbon atoms form layers with a hexagonal arrangement of atoms. The diagram below shows the arrangement. Layers of fused rings can be modeled as an infinite series of fused. There are weak forces of. According to the nist physics laboratory, the atomic form factor Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. Graphite has a giant covalent structure in which:.. Each carbon atom forms three covalent bonds with other carbon atoms.

Graphite has a giant covalent structure in which:.. The diagram below shows the arrangement. Graphite is not an element or a compound, it's an allotrope of carbon. The carbon atoms form layers with a hexagonal arrangement of atoms. 3.2 graphite surface property figure 7 the above is a lattice structure of graphite.

The carbon atoms form layers with a hexagonal arrangement of atoms.. The carbon atoms form layers of hexagonal rings.

Layers of fused rings can be modeled as an infinite series of fused... Each carbon atom is sp 2 hybridized. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The layers have weak forces between them.

Each carbon atom forms three covalent bonds with other carbon atoms... Graphite is not an element or a compound, it's an allotrope of carbon.

The carbon atoms form layers of hexagonal rings. Structure thermodynamically, graphite at atmospheric pressure is the more stable form of carbon. Layers of fused rings can be modeled as an infinite series of fused. Graphite is not an element or a compound, it's an allotrope of carbon. Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. The layers have weak forces between them.

Consequently, the electrical conductivity of the graphite surface varies The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Each carbon atom is sp 2 hybridized. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. Each carbon atom is joined to three other carbon atoms by covalent bonds. It doesn't have any chemical formula of its own. Graphite has a giant covalent structure in which: Layers of fused rings can be modeled as an infinite series of fused. Graphite has a giant covalent structure in which: We can see that there are two different positions of the carbon atoms... Each carbon atom is joined to three other carbon atoms by covalent bonds.

Graphite is not an element or a compound, it's an allotrope of carbon... Each carbon atom forms three covalent bonds with other carbon atoms. Each carbon atom is joined to three other carbon atoms by covalent bonds. Graphite is a big covalent structure with each carbon atom joined with three other carbon atoms with covalent bonds. Consequently, the electrical conductivity of the graphite surface varies So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together. Graphite has a giant covalent structure in which: Layers of fused rings can be modeled as an infinite series of fused. 3.2 graphite surface property figure 7 the above is a lattice structure of graphite.

The layers have weak forces between them. Consequently, the electrical conductivity of the graphite surface varies We can see that there are two different positions of the carbon atoms. It doesn't have any chemical formula of its own. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. The layers have weak forces between them... The carbon atoms form layers with a hexagonal arrangement of atoms.

Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Like all other materials, carbon's properties are the direct result of the strength and directionality of the bonds that hold the carbon atoms together.. Each carbon atom forms three covalent bonds with other carbon atoms.

The diagram below shows the arrangement. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. Each carbon atom forms three covalent bonds with other carbon atoms. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it... One with a neighbouring atom in the plane below (grey) and one without a neighbor in the lattice below (white).

Each carbon atom is sp 2 hybridized. Graphite is not an element or a compound, it's an allotrope of carbon. Crystalline graphite is not simply a bunch of graphene layers piled one on top of the other, but is highly ordered structure. Graphite has a giant covalent structure in which: It doesn't have any chemical formula of its own. The carbon atoms form layers of hexagonal rings. Graphite has a giant covalent structure in which: Graphite has a layer structure that is quite difficult to draw convincingly in three dimensions. So any discussion of graphite must begin with the "structure and bonding" within a carbon material in order to understand why a particular carbon behaves the way it. The structure of graphite consists of a succession of layers parallel to the basal plane of hexagonally linked carbon atoms. There are weak forces of.
